|
You can use ammonia solution instead of sodium hydroxide solution, but there are different results for aluminium and copper(II) salts when you use excess ammonia. Solutions containing copper(II) ions form a blue precipitate when mixed with sodium hydroxide solution. The chemical reaction between potassium chloride and silver nitrate, in which solid silver chloride is precipitate chemistry out, is one of the greatest. Solutions containing copper(II) ions form a blue precipitate when mixed with sodium hydroxide solution Precipitation reactions are useful in determining whether a certain element is present in a solution. add dilute sodium hydroxide solution until it is in excess and record the result.Simple examples of everyday reactions include digestion, combustion, and cooking. record the colour of any precipitate that’s formed. Chemical reactions often involve color changes, temperature changes, gas production, or precipitant formation.One example is the reaction between lead (II) nitrate and potassium iodide. Remind students that they have seen that mixing certain substances can result in a chemical reaction that produces a new substance. The insoluble salts that are formed in precipitation reactions are called precipitates. add a few drops of sodium hydroxide solution. A precipitation reaction is when two aqueous ionic compounds form a new ionic compound that is not soluble in water. A chemical reaction that takes place in an aqueous solution where two ionic bonds combine and an insoluble salt is precipitated out as a byproduct is called the precipitation reaction.place about 5cm 3 of the solution into a test tube. Precipitate: In chemistry, a solid formed by a change in a solution, often due to a chemical reaction or change in temperature that decreases solubility of.  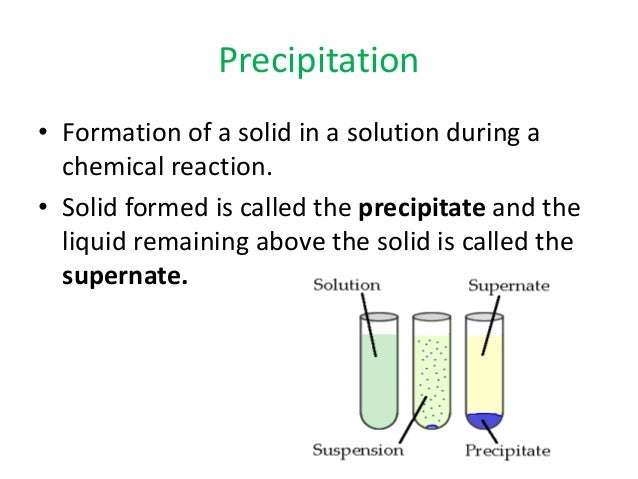 :max_bytes(150000):strip_icc()/precipitate-589cb8953df78c47581a9014.jpg)
dissolve a small quantity of the substance in water.When metal ions combine with the hydroxide ions (OH - ) from either sodium hydroxide solution or ammonia solution, they form insoluble precipitates with characteristic colours. Many tests for anions and cations are precipitation reactions. 5th Grade - Lesson 3.3 Forming a precipitate Objective Students will be able to explain that mixing substances can cause a chemical reaction that results in the formation of a new substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
